
People using this therapy need to continue working with others on their health care team. Spinal cord stimulation interferes with pain signals sent to the brain, but does not treat the underlying condition. The device should be turned off and the doctor contacted if this occurs. Unwanted changes in stimulation may include a jolting or shocking feeling. Less serious disadvantages of spinal cord stimulation devices include: The appropriate use of neurostimulation of the spinal cord and peripheral nervous system for the treatment of chronic pain and ischemic diseases: the Neuromodulation Appropriateness Consensus Committee. While complications related to the implanted devices are relatively common, they appear to be declining with advances in technology and surgeons’ skills.Ĥ Deer TR, Mekhail N, Provenzano D, et al. The most common complications were unintended movement (also called migration) of the leads, failed connections in leads, and breakage of leads. Retrospective review of 707 cases of spinal cord stimulation: indications and complications. One extensive study in the medical literature found 38% of the research participants had device-related problems.ģ Mekhail NA, Mathews M, Nageeb F, Guirguis M, Mekhail MN, Cheng J. “Together with the compatibility of multiple types of waveform options, inclusive of DTM SCS, and industry-leading MRI access, I believe this latest generation of rechargeable neurostimulator represents a significant leap forward in SCS therapy and a milestone of modern neuromodulation.Spinal cord stimulation and peripheral nerve field stimulation are considered generally safe, with the potential risks mainly related to the surgical procedures required for a trial period or long-term therapy. The Inceptiv closed-loop feature will allow for a more consistent and optimal delivery of therapy, and reduced need for manual programming adjustments by the patient. “This can compromise the patient´s experience and therapeutic effectiveness. Dirk Rasche of the Department of Neurosurgery, University Hospital of Schleswig-Holstein in Lübeck, Germany. 
“Today, fixed-output spinal cord stimulation does not account for patient movements, which can alter the distance between the spinal cord and implanted epidural leads,” said Dr. It remains unapproved for sale or distribution in the U.S. Medtronic plans to make Inceptiv commercially available in the coming months in Europe. Its rechargeable battery can go from empty to full in approximately one hour, too. Most people using SCS need an MRI within five years of receiving their device, the company said.Īdditionally, Inceptiv is the thinnest SCS device in the world at 6 mm, the company said. 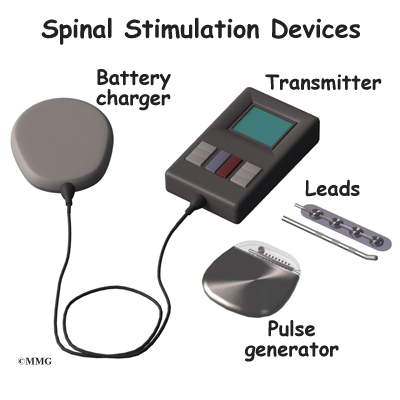
Medtronic said its device is the only SCS system in Europe offering both full-body 1.5T and 3T MRI access. “This approval marks the beginning of a new era of pain relief, using sensing technology to listen to the unique biological signals of each patient.” More about the Medtronic Inceptiv system Ash Sharan, chief medical officer of the Neuromodulation unit at Medtronic. “Medtronic pioneered SCS for pain management more than 50 years ago, and we continue to deliver new innovations that personalize care and improve pain relief for patients,” said Dr. A clinical study showed that nine of 10 patients preferred the closed-loop option to fixed-output stimulation. The closed-loop technology unlocks the ability to listen and respond to signals along the spinal cord. Inceptiv utilizes a closed-loop capability with ECAPs (evoked compound action potentials) based on decades of Medtronic research. All this occurs seamlessly while patients perform a full range of daily activities. As neural response decreases, stimulation returns to the pre-set, optimal level. When neural response increases - for instance, during a cough or sneeze - Inceptiv automatically decreases stimulation. That, in turn, can lead to patients turning down their device’s stimulation output, negating their therapy.Ĭonversely, Inceptiv senses the body’s neural response to stimulation 50 times per second, every second, every day. Medtronic says that keeps therapy in harmony with the motions of daily life.Īccording to a news release, certain movements during SCS therapy may result in uncomfortable stimulation. This feature senses each person’s unique biological signals, adjusting stimulation moment-to-moment as needed. Medtronic said it’s the company’s first SCS device to offer a closed-loop feature. Inceptiv, a closed-loop, rechargeable SCS, treats chronic pain. + announced today that it received CE mark approval for its Inceptiv spinal cord stimulator (SCS). The Inceptiv spinal cord stimulation system.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
